We can now look at the bonding in ammonia, NH 3. Orbitals puts the electron density directly between the nuclei. The overlap of the hydrogen s orbitals and the carbon sp 3 S) because they are formed from hybridized orbitals, which result from s orbitals. These will pair up with the carbon electrons to form four σ(sigma) bonds. Each of the hydrogens has one valenceĮlectron in its 1 s orbital ( ↓). The four valence electrons on carbon can beĪdded to the energy diagram (↑). Hybridization, all four hybrid orbitals have the same energy, lower than p orbitals, but higher than s orbitals. Hybridization also changes the energy levels of the orbitals. Orbitals to give the predicted tetrahedral geometry and shape of methane, The molecular, sp 3 orbitals are arranged in a tetrahedron, The shape and orientation of these new molecular orbitals are shown In carbon, the atomic wave functions, Ψ, for the 2 sĪnd three 2 p orbitals are mixed mathematically, to give new molecular wave functions, and molecular orbitals. There are only two p orbitals in the carbon atoms that have 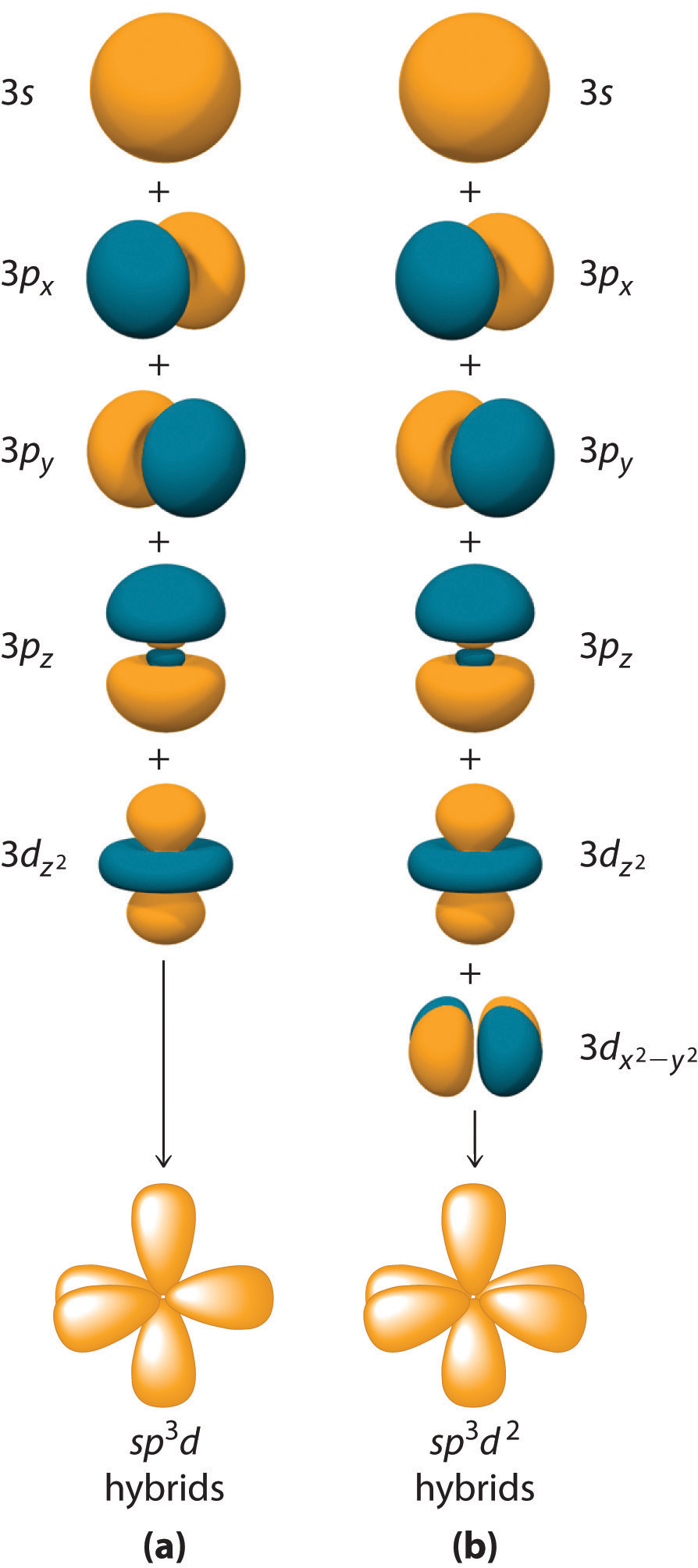
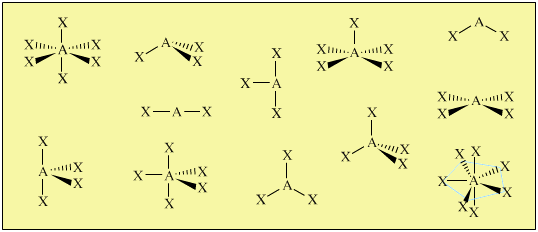
Hydrogen atoms have the electron configuration 1 s 1.Ĭarbon atoms have the electron configuration 2 s 2 2 p 2.Ĭontribute 1 electron to a C-H bond. Shape, based on the four atoms bonded to the central atom. In the case of H 2, the 1 s orbitals, each containing one electron, overlap to form the H-H To concentrate the electron density between atoms to form chemicalīonds. Lewis theory proposed that chemical bonds form whenīond theory describes how the orbitals of bonding species merge (overlap) What this model does notĮxplain is the nature of chemical bonding, the chemical properties of We can use VSEPR to predict molecular geometries and Which only interacts with other polar species, "likes dissolve In CCl 4, the four polar C-Cl bonds willĬancel out, making this a non-polar molecule. In the CHCl 3 molecule, the three polar C-Cl bondsĪdd (vector addition) to give a net dipole moment to the molecule. They will both have tetrahedral geometries, with 4 (For a similar example, draw the Lewis structures of BF 3Īnother indication of the importance of molecular shape canīe seen by comparing the physical properties of CHCl 3 and CCl 4. They will, in fact add, and give a netĬompound, which explains (as we will soon see) its This will give it a bentĭipoles are not pointing in opposite directions, and will not cancel Its molecular geometry will be trigonalīecause of the three valence electron pairs on sulfur, two bonding pairsĪnd a lone pair. Shown below is the shape of an SO 2 molecule. Is a non-polar compound, due to its molecular shape. Molecular shape of CO 2, they are pointing in opposite directions, It will have the shape shown below.Īs indicated by the arrows, there are two very polar bonds in There are only two electron pairs on the central carbon atom. Than just the presence of the two polar bond in each Properties, CO 2 boils at -78 oC, and SO 2īoils at +22.8 oC, a 100 odifference. However, they have very different physical The overall compound will be determined by the presence of polar bonds andįor example, we can compare carbon dioxide, CO 2Ĭarbon and sulfur have the same electronegativity, much less In larger molecules (more than two atoms), the polarity of (melting and boiling points, solubilities, etc.). This will affect their physical properties All diatomic molecules containingĪtoms of different electronegativities will be polar molecules. This indicates that the H will carry a partial positiveĪnd the F will carry a partial negative charge (δ -). Usually indicated by the presence of an arrow, as shown below for HF. The direction of a dipole moment (charge imbalance) is 
Hydrogen and the shared pair of bonding electrons will spend more time near the Fluorine is much more electronegative than This will arise from polar bonds within the molecule, due toĭifferences in electronegativity values between bonded atoms. Polarity exists when there is a separation Once we know a molecular shape, we can start to look at the physicalĮxample, we should now be able to predict which molecules will be polar. We can then use VSEPR to predict molecular shapes, based on the valenceĮlectron pairs of the Lewis structures. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
